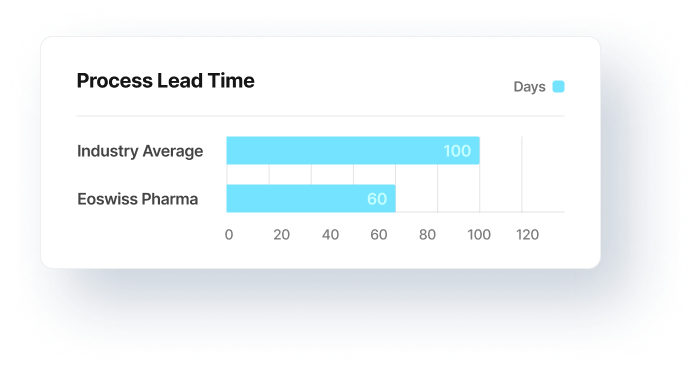
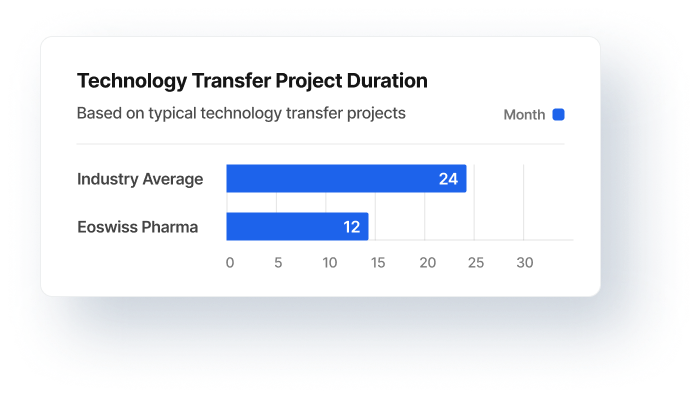
GMP-compliant PAT (Process Analytical Technology) implementation and process monitoring. Solid data integration for production, quality and purchasing as a basis for decision-making. With 30% reduced lead times to release. This is how they efficiently avoid data waste at reasonable costs.

SMEs and large corporations
from Switzerland and Europe
Projects in Switzerland – EU – China – Japan – USA
MSAT & Manufacturing & Quality Supply Chain & Regulatory
SMEs and large corporations
from Switzerland and Europe
Projects in Switzerland – EU – China – Japan – USA
MSAT & Manufacturing & Quality Supply Chain & Regulatory
Programme Management
Solid data modelling, scale up
Technically very demanding
US Corporation
Project Management
50% deeper transfer costs and a more stable production process with process design.
Problem: little data
Client: Group France
Project Management
Networked IR Camera System
pattern recognition
IT Connectivity
Customer: Pharmaceutical SMEs EU
Many of these costs remain unnoticed for a long time, but they have a direct impact on costs and availability.
Problems only become apparent when projects are already delayed.
Fehlende Daten müssen vor der Entscheidung erst beschafft werden. Das führt zu Verspätungen und verspätetem Umsatz
Inefficient production
Small changes must be laboriously tested instead of being simulated. This costs time, money, and leads to discussions.
Poor digitalisation leads to long lead times for approval. This results in less work being invoiced.
Missing data must be obtained before the decision can be made. This leads to delays and delayed revenue.
Inefficient production
Small changes must be laboriously tested instead of being simulated. This costs time, money, and leads to discussions.
Poor digitalisation leads to long lead times for approval. This results in less work being invoiced.
No question of IT or software
Digitalisation projects start euphorically, but then often lose considerable momentum.
These are often the problems:
Digitalisation doesn't need giant projects but rather robust, practical, step-by-step implementation. This reduces costs and reduces risk.

We believe that practical digitalisation (outside the „buzzword sphere“) makes the difference between profit and loss.
We develop our own software along with sensors. 15% of our revenue goes into research and development.
For swift process decisions. With our tailor-made models, we optimise validation parameters and batch sizes for cost minimisation and GMP safety.
The practical application of Statistical Process Control (SPC) along with systematic improvement makes the manufacturing process safe and faster.
DoE and Taguchi optimisations.
“Process Analytical Technology” often means expensive software licences and complicated IT suppliers.
We build custom-made models, without the ballasts. With up to 30 % lower costs.
Adapt processes and workflows before actual digitisation. This ensures that business processes are future-proof for the coming years.
Our experienced team will help you implement this easily.
Media breaks practically invite expensive rework. Delivery notes, quality certificates, batch certificates, etc. often exist in different forms.
Processes on a single channel, reducing throughput time and costs
Many databases are created, but no one feels responsible for data maintenance.
Unmaintained databases create organisational duplications that cost time and money.
Databases need to be cleaned up.
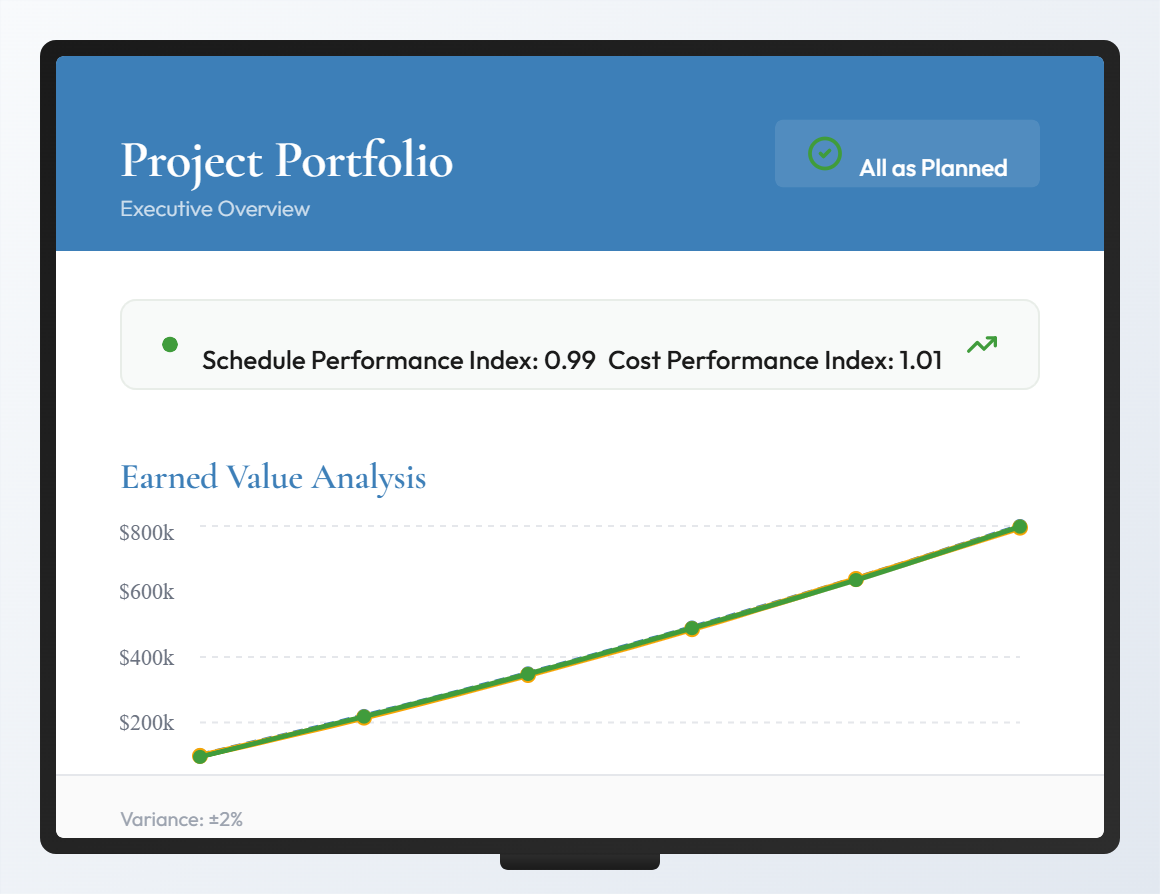
We define goals for each project, the achievement of which we also measure. We can do this because we implement our proposals ourselves, rather than just advising. Our improvements are measurable, verifiable, and demonstrable. This is what sets us apart from large, purely consulting firms.
We have operational management experience and technical experience. Our implementations are presentable and GMP compliant.
We specialise in implementations. We know that a technical solution must also be anchored as an administrative process, otherwise it won't work.
Here are some of the important advantages
For our project implementation, there are prerequisites that need to align.
Our methods are not suitable for resolving conflicts between customers and suppliers.
We can facilitate conflict resolution, but we do not conduct it ourselves.
Various restructurings are already underway in parallel. Then we cannot conduct a realistic measurement of success.
We always measure success in our implementations. Once for the set project plan and budget, but we also pursue internal goals: for example, the implementation and introduction of a new technique during project execution. If too many projects are running in parallel, we can no longer measure and manage the success of our projects.
Let's have a brainstorm here.
When you ask this question, they've probably already had unpleasant
Experienced surprises.
Our project costs are billed based on time and effort. We provide an initial estimate of time and costs as early as the first meeting. This budget framework also works out. The overall costs are lower the faster we are allowed to be.
Cost transparency and cost control after
Project times and phases
Why do we make our projects better? We are experienced specialists in our areas of focus. Large consulting firms have neither this knowledge nor this experience.
We actively conduct our own research, keeping us at the cutting edge.
These are the processes and tools we use the most
Scale-up and process development.
Bespoke software development for tailored solutions
Development of freeze-drying cycles. Scale up & Scale down. Technology transfer of freeze-drying.
Scale-up and process development. Modelling of processes with mathematical and statistical models.
Optimisation and Process Development
Design of Experiments (DoE), optimisation of plants during ongoing production, process simulations
Process Analytical Technology (PAT)
Linking experimental data to models, using these models for process development and optimisation
Finding robust production parameters. This is where manufacturing profitability lies.
Quality Management under GMP (Pharmaceuticals, Medical Technology, Chemicals, Industry) or ISO 9001, ISO 13485
We are dealing with the optimisation of warehouses, routes, buffers for future extensions and conversions.
Our project management also includes change management. This is the only way to achieve sustainable implementation.
Without implementation, no project helps.
Transfer of technologies, productions, expansion and refurbishment of facilities
Transfer of analytical methods under GMP, including validations, verifications with all necessary documentation
Professional process optimisations (in collaboration with Japanese specialists).
These are the processes and tools we use the most
Scale-up and process development.
Bespoke software development for tailored solutions
Development of freeze-drying cycles. Scale up & Scale down. Technology transfer of freeze-drying.
Scale-up and process development. Modelling of processes with mathematical and statistical models.
Optimisation and Process Development
Design of Experiments (DoE), optimisation of plants during ongoing production, process simulations
Process Analytical Technology (PAT)
Linking experimental data to models, using these models for process development and optimisation
Finding robust production parameters. This is where manufacturing profitability lies.
Quality Management under GMP (Pharmaceuticals, Medical Technology, Chemicals, Industry) or ISO 9001, ISO 13485
We are dealing with the optimisation of warehouses, routes, buffers for future extensions and conversions.
Our project management also includes change management. This is the only way to achieve sustainable implementation.
Without implementation, no project helps.
Transfer of technologies, productions, expansion and refurbishment of facilities
Transfer of analytical methods under GMP, including validations, verifications with all necessary documentation
Professional process optimisations (in collaboration with Japanese specialists).
Digitalisierungsprojekte in der Pharma-Herstellung scheitern häufig, weil sie als IT-Projekte behandelt werden und nicht an der GMP-Realität der Produktion ausgerichtet sind. Typische Probleme sind unklare Prozessziele, mangelnde Einbindung von QA, MSAT und Herstellung sowie Daten, die im Alltag nicht nutzbar sind. Dadurch steigen Komplexität und Compliance-Risiken, ohne dass die Performance messbar besser wird.
Pharma-Herstellung lässt sich sicher modernisieren, wenn Prozesse zuerst verstanden, vereinfacht und anschließend digital unterstützt werden. Wichtig sind klare GMP-Anforderungen, Datenintegrität, frühe QA-Einbindung und eine praktische Umsetzung in der Produktion. So entsteht Digitalisierung, die Performance verbessert und gleichzeitig auditfähig bleibt.
Daten und PAT helfen, kritische Prozessparameter besser zu verstehen, Abweichungen früher zu erkennen und Entscheidungen auf eine verlässliche Grundlage zu stellen. Entscheidend ist jedoch, dass Daten nicht nur gesammelt, sondern für konkrete Prozessverbesserungen genutzt werden. Nur dann verbessern Digitalisierung und PAT tatsächlich Effizienz, Qualität und GMP-Sicherheit.
Die Performance in der Pharma-Herstellung verbessert sich durch transparente Prozessdaten, klare Kennzahlen und gezielte Optimierung kritischer Engpässe. Dazu gehören stabilere Abläufe, weniger manuelle Arbeitsschritte, bessere Prozesskontrolle und eine engere Abstimmung zwischen Herstellung, QA und MSAT. Der Nutzen muss an messbaren Ergebnissen wie Durchlaufzeit, Abweichungen, Freigabezeiten und Kosten sichtbar werden.
Externe Experten sind sinnvoll, wenn Digitalisierungsprojekte nicht die erwarteten Ergebnisse liefern, die Komplexität steigt oder Compliance-Bedenken entstehen. Besonders bei überlasteten internen Teams können erfahrene GMP- und Produktionsexperten helfen, den richtigen Fokus zu setzen, Risiken zu reduzieren und die Umsetzung zu beschleunigen.