Why do pharmaceutical projects take a long time?
Why are GMP projects delayed?
GMP-compliant implementation strategies that shorten critical project paths for Tech Transfer, Scale Up and QA projects by 30-40%% while ensuring full regulatory control

SMEs and large corporations
from Switzerland and Europe
Projects in Switzerland – EU – China – Japan – USA
MSAT & Manufacturing & Quality Supply Chain & Regulatory
Programme Management
Construction of production buildings
High time pressure, complex logistics
15 months instead of 19 months
(Brownfield / Greenfield Project) Client: DAX Corporation
Overall Project Management
Emergency Production Transfer
Extreme time pressure
6 months instead of 18 months
Customer: Pharmaceutical SMEs
Programme Management
Complex coordination new factory construction, time pressure
Customer: Dax Group
Production building
(Brownfield / Greenfield Project) DAX Corporation
Emergency Production Transfer
Pharmaceutical SMEs; Germany
Our biggest project: New factory construction for Dax Group
Unmet quarterly figures generate unnecessary discussions
Additional capacity for the delayed project, slowing down other activities
Additional staff cause unnecessary repetitions and unplanned additional costs.
Production building
(Brownfield / Greenfield Project) DAX Corporation
Emergency Production Transfer
Pharmaceutical SMEs; Germany
Our biggest project: New factory construction for Dax Group
No question of IT or software
Projects are very rarely delayed due to the wrong software or insufficient IT infrastructure. These are often these problems:
If these problems are not resolved, project times and costs will double
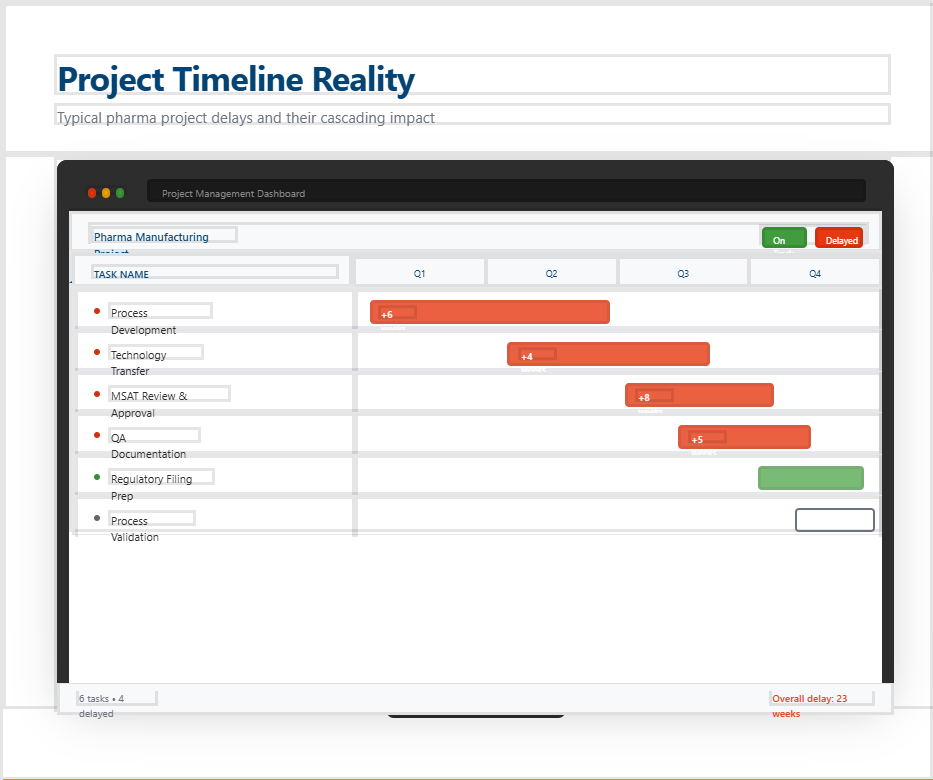
Assets are not directly comparable.
Detail adjustments are needed.
Further coordination between QA, Production and the client is necessary.
The already long lead times for components are being exceeded
Production building
(Brownfield / Greenfield Project) DAX Corporation
Emergency Production Transfer
Pharmaceutical SMEs; Germany
Our biggest project: New factory construction for Dax Group
In our experience, technical and technological obstacles are very rarely the real problem areas of projects and processes.
There are numerous theories on project management which can easily be read up on. Implementation, on the other hand, is learned over the years. Only solid implementation brings real cost advantages.
With a few months of external support, you will reorganise the projects and achieve very fast, and therefore cost-effective, implementation.
External experts build on their experience. This allows processes to be specifically improved while simultaneously reducing GMP risk.
We conduct optimisations as simulations, based on measurement data and laboratory tests. This reduces the cost to 10 % of production trials.
When SOPs no longer work, we overhaul them, streamline and simplify them. This is how a simple document change improves the economic result.
Technology and engineering are aligned from the outset. This allows complex technology transfers to be carried out simply and safely.
In the technical field, it is easier to measure process performance. However, it is more important in administrative processes in areas such as Quality, Regulatory, and Business Development. Measurement is necessary in order to improve.
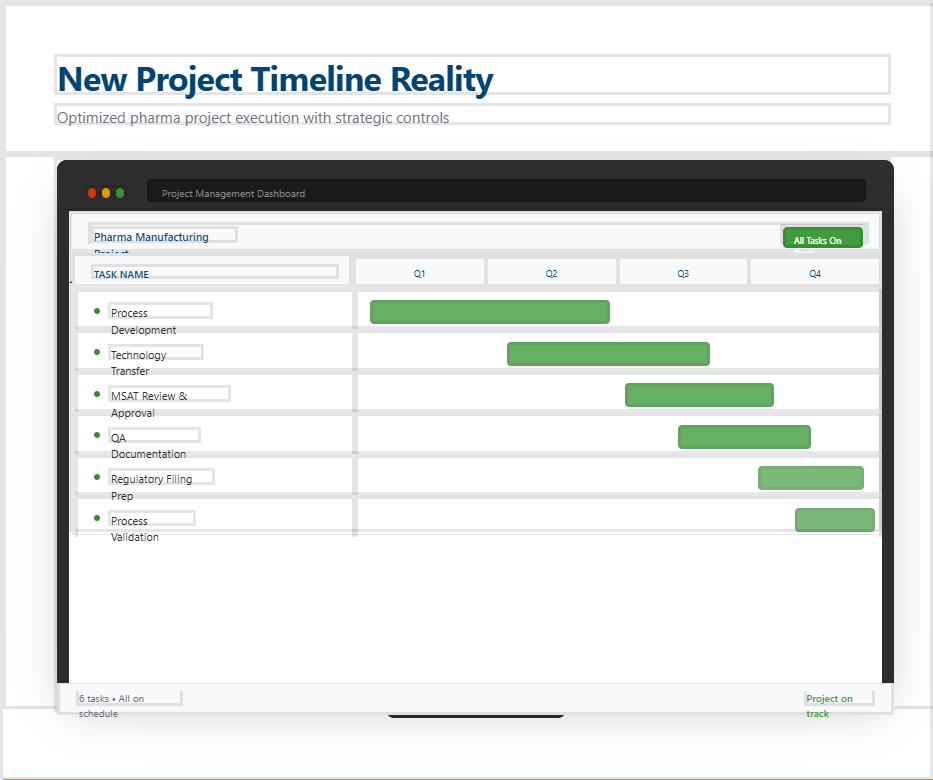
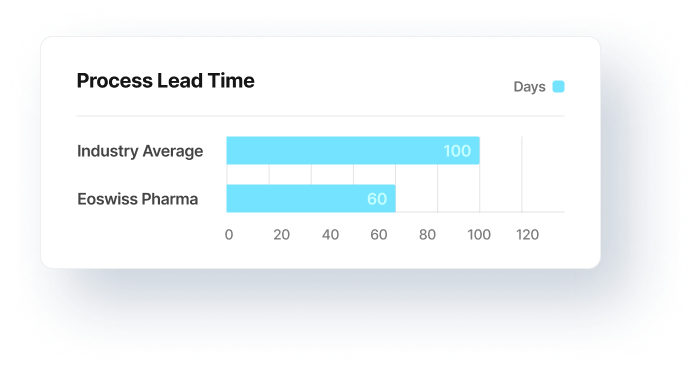
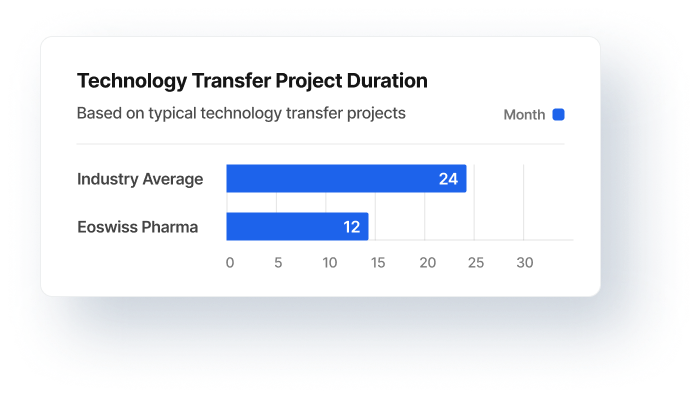
The total duration of a project (e.g. technology transfers) determines the overall costs of projects and business processes.
The project duration directly determines costs, revenue, and capacity utilisation.
Here are some of the important advantages
For our project implementation, there are prerequisites that need to align.
Our methods are not suitable for resolving conflicts between customers and suppliers.
We can mediate conflict resolution, but we do not conduct it ourselves.
Various restructuring efforts are already underway in parallel. Then we cannot carry out a realistic measurement of success.
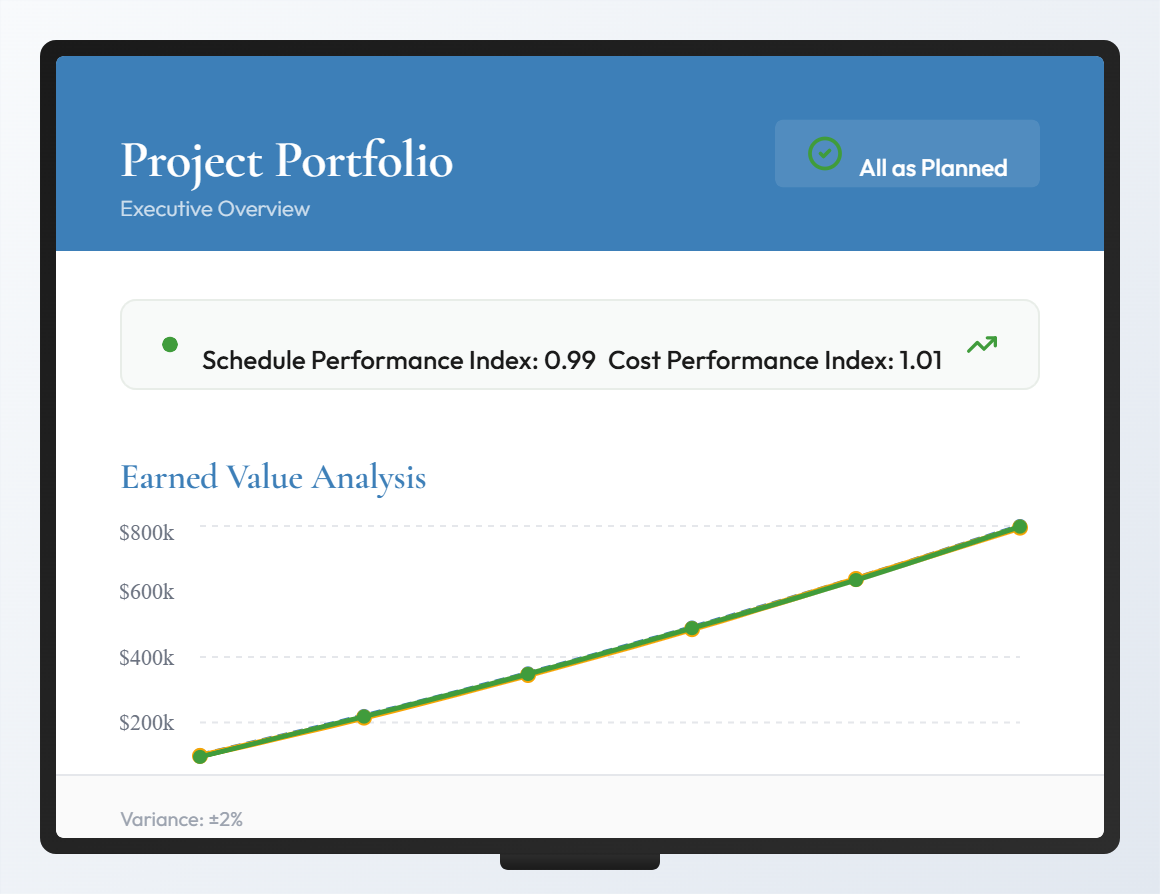
We always carry out a success measurement for our implementations. Once regarding the set project plan and budget, but we also pursue internal goals: determining our own lead times and implementation times, for example. If too many projects are running in parallel, we can no longer track and manage the success of our project.
Let's have a brain-dump on that.
If you are asking this question, you have probably already experienced unpleasant surprises.
Our project costs are billed based on time and effort. We provide an initial estimate of time and costs right from the first meeting. This budget framework also works. The overall costs are lower the faster we are allowed to be.
Cost transparency and cost control after
Project times and phases
Why do we make our projects better? We are experienced specialists in our areas of focus. Large consulting firms have neither this knowledge nor this experience.
We actively conduct our own research, keeping us at the cutting edge.
These are the processes and tools we use the most
Scale-up and process development.
Bespoke software development for tailored solutions
Development of freeze-drying cycles. Scale up & Scale down. Technology transfer of freeze-drying.
Scale-up and process development. Modelling of processes with mathematical and statistical models.
Optimisation and Process Development
Design of Experiments (DoE), optimisation of plants during ongoing production, process simulations
Process Analytical Technology (PAT)
Linking experimental data to models, using these models for process development and optimisation
Finding robust production parameters. This is where manufacturing profitability lies.
Quality Management under GMP (Pharmaceuticals, Medical Technology, Chemicals, Industry) or ISO 9001, ISO 13485
We are dealing with the optimisation of warehouses, routes, buffers for future extensions and conversions.
Our project management also includes change management. This is the only way to achieve sustainable implementation.
Without implementation, no project helps.
Transfer of technologies, productions, expansion and refurbishment of facilities
Transfer of analytical methods under GMP, including validations, verifications with all necessary documentation
Professional process optimisations (in collaboration with Japanese specialists).
These are the processes and tools we use the most
Scale-up and process development.
Bespoke software development for tailored solutions
Development of freeze-drying cycles. Scale up & Scale down. Technology transfer of freeze-drying.
Scale-up and process development. Modelling of processes with mathematical and statistical models.
Optimisation and Process Development
Design of Experiments (DoE), optimisation of plants during ongoing production, process simulations
Process Analytical Technology (PAT)
Linking experimental data to models, using these models for process development and optimisation
Finding robust production parameters. This is where manufacturing profitability lies.
Quality Management under GMP (Pharmaceuticals, Medical Technology, Chemicals, Industry) or ISO 9001, ISO 13485
We are dealing with the optimisation of warehouses, routes, buffers for future extensions and conversions.
Our project management also includes change management. This is the only way to achieve sustainable implementation.
Without implementation, no project helps.
Transfer of technologies, productions, expansion and refurbishment of facilities
Transfer of analytical methods under GMP, including validations, verifications with all necessary documentation
Professional process optimisations (in collaboration with Japanese specialists).
Pharma projects can be implemented faster if bottlenecks in execution are identified early and addressed directly. Clear responsibilities, close coordination between MSAT, QA, and production, and active management of CDMOs are crucial. This significantly reduces delays in technology transfer and validation.
GMP projects are frequently delayed due to overloaded MSAT and QA teams, a lack of coordination between departments, and insufficient oversight of external partners like CDMOs. These factors lead to delays in technology transfer, validation, and batch release, whilst simultaneously increasing compliance risk.
GMP risks can be reduced if quality requirements are integrated into the implementation from the outset. This includes close coordination between QA, MSAT, and manufacturing, as well as consistent documentation throughout the real process. Early identification of deviations prevents problems during audits and inspections.
Collaboration with CDMOs improves through clear governance structures, transparent communication, and active management of external partners. It is important that progress, risks, and responsibilities are continuously monitored to avoid delays and quality issues.
External experts are useful when projects are delayed, validations fail, or internal teams are overloaded. They help to quickly identify bottlenecks, accelerate decision-making, and bring projects back under control.
Technology transfer can be accelerated by clear process definitions, early alignment between MSAT, QA and production, and structured management of CDMOs. It is crucial to identify and actively manage risks early on, rather than reacting to them.