We help Pharma Management teams to permanently improve contract manufacturers (CMOs) and strengthen supply chains. All this while adhering to all GMP rules and without any risk to existing partnerships.

SMEs and large corporations
from Switzerland and Europe
Projects in Switzerland – EU – China – Japan – USA
MSAT & Manufacturing & Quality Supply Chain & Regulatory
Project Management
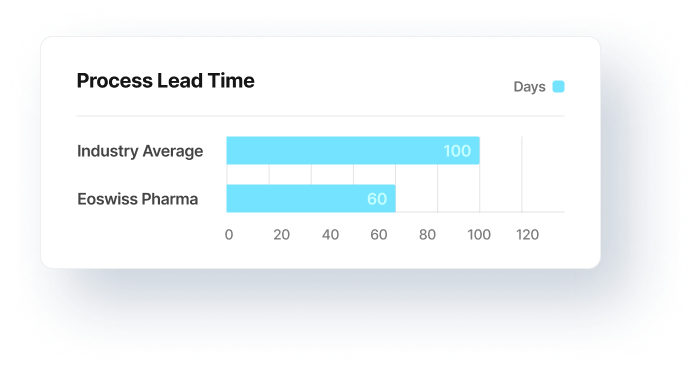
Doubled performance
critical supplier
Problem: non-robust CMOs
Client: Corporation; Switzerland
Project Management
Variable batch sizes: No more inventory write-offs
1 batch per year
Customer: Pharmaceutical SMEs
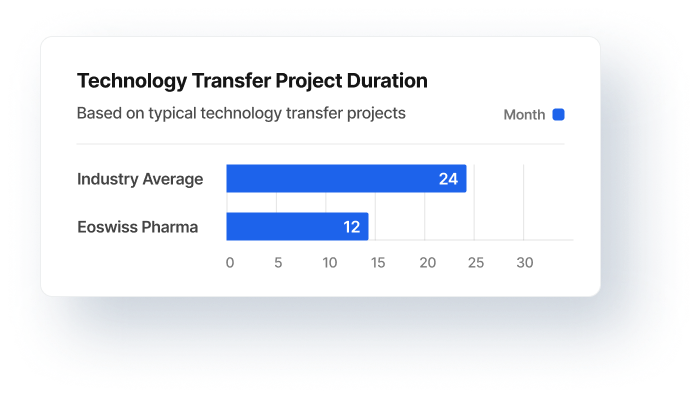
Programme Management
Technology transfer, production transfer
to outsourcing partners
Establishment of CMOs
Customer: Pharmaceutical SMEs EU
Doubled performance
critical supplier
(International conglomerate; Switzerland)
Variable batch sizes: no stock write-offs
more
Pharmaceuticals Medium-Sized Enterprises; Germany
Technology
producer, production transfer
to outsourcing partners
(Pharmaceutical SMEs EU)
Large batch sizes at „favourable“ costs lead to inventory write-downs because consumption can never be predicted precisely.
You can be easily pressured. Ways out of the supplier bottleneck cost money and nerves.
Even in the pharmaceutical industry, there are trends, for example, in product presentation. With a fast supply chain, you gain market share. .
Production building
(Brownfield / Greenfield Project) DAX Corporation
Emergency Production Transfer
Pharmaceutical SMEs; Germany
Our biggest project: New factory construction for Dax Group
Most of the time, it's not one single problem, but rather performance eroding due to a variety of inefficiencies:
70% der Wertschöpfung findet in der Lieferkette statt – nur 30 % in der eigenen Herstellung. A strong supply chain pays off financially.
Resolve these issues immediately:
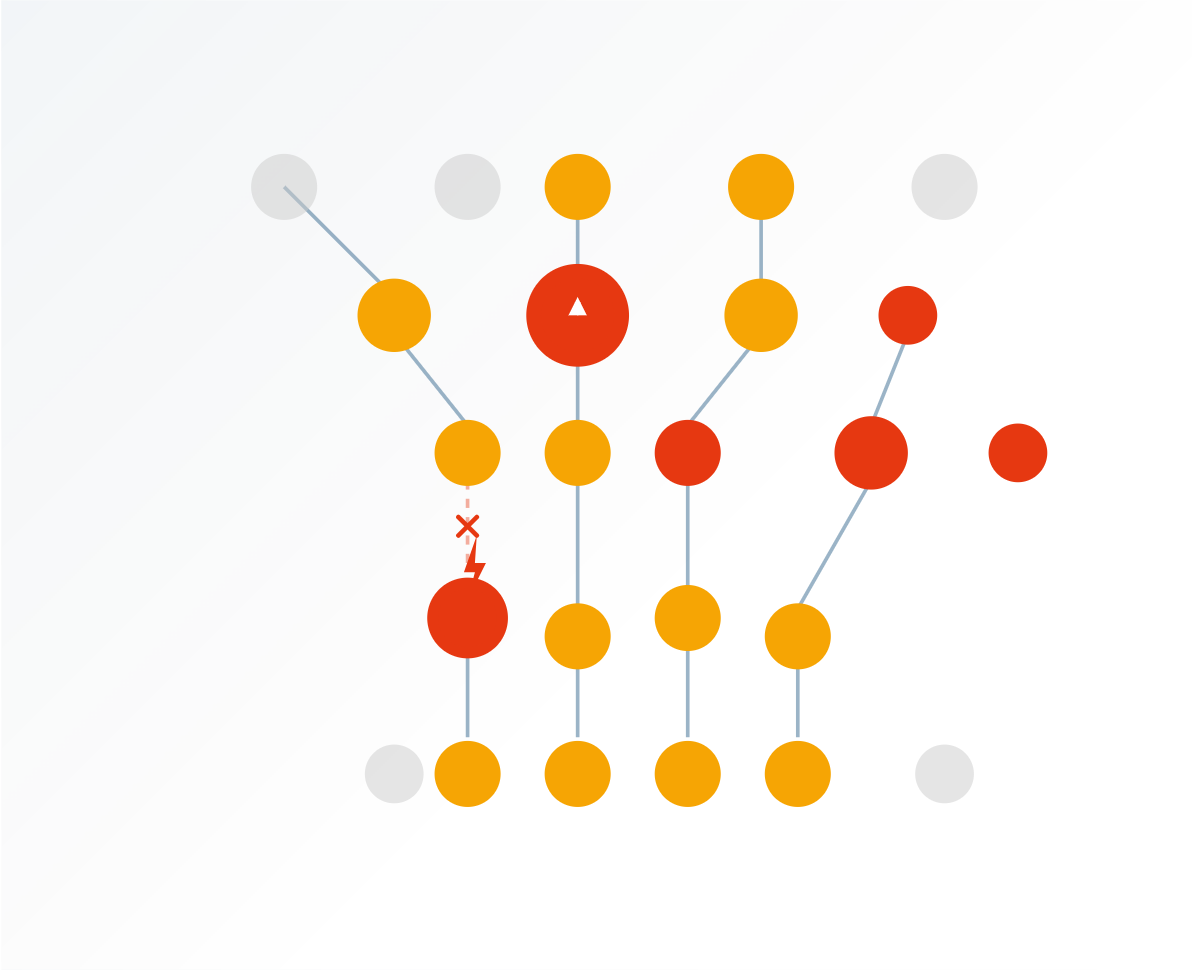
In our experience, there isn't usually one specific problem. Most of the time, the performance of contract manufacturers erodes due to several inefficiencies. We prioritise eliminating these in our projects, thereby improving performance.
China and India are important hubs. However, depending on the product, Europe, Japan, Korea or the USA could be cheaper.
We are implementing your project according to plan and are geographically experienced. You will receive a robust, resilient and cost-effective supply chain.
Our improvements are based on measured performance. To achieve this, we operate a transparent monitoring system.
Our process selects for the overall performance of a manufacturer or supply chain, not just KPIs.
Business processes at the CMO do not have the same objective as the customer's business processes.
We are carrying out a synchronisation of these goals. Processes will become faster and more robust.
Ideal batch sizes have to be constantly readjusted to react to the market.
We support these changes. This ensures you always have a competitive cost structure.
New and existing manufacturing partners are analysed to achieve the ideal mix of manufacturing capabilities.
We will find the ideal outsourcing partner and manage the entire transfer.
You will receive higher COGS.
We analyse the manufacturer with the production process and establish process improvements.
Our monitoring data and KPIs highlight the capable suppliers.
The performance of the supply chain is defined and continuously measured. We do not measure with a multitude of KPIs, but prefer meaningful metrics.
Our improvements are measurable, demonstrable, and presentable.
The total costs of manufacturing over the entire year include production costs, but also quality issues and their costs, stock-outs due to late deliveries, inventory write-downs, and re-planning for CMO and sales.
A large proportion of the costs are “hidden costs.” We identify these costs and make them visible. Then we systematically reduce these cost blocks. Through technical optimisations and adjustments in the order- delivery- invoicing process.
Here are some of the important advantages
For our project implementation, there are prerequisites that need to align.
Our methods are not suitable for resolving conflicts between customers and suppliers.
We can mediate conflict resolution, but we do not conduct it ourselves.
Various restructurings are already running in parallel. Then we cannot carry out realistic success measurement.
We always carry out a success measurement for our implementations. Firstly, according to the set project plan and budget, but we also pursue internal goals: determining our throughput times and implementation times, for example. If too many projects are running in parallel, we can no longer measure and manage the success of our projects.
Let's have a brainstorm here.
When you ask this question, they've probably already had unpleasant
Experienced surprises.
Our project costs are billed based on time and effort. We provide an initial estimate of time and costs as early as the first meeting. This budget framework also works out. The overall costs are lower the faster we are allowed to be.
Cost transparency and cost control after
Project times and phases
Why do we make our projects better? We are experienced specialists in our areas of focus. Large consulting firms have neither this knowledge nor this experience.
We actively conduct our own research, keeping us at the cutting edge.
These are the processes and tools we use the most
Scale-up and process development.
Bespoke software development for tailored solutions
Development of freeze-drying cycles. Scale up & Scale down. Technology transfer of freeze-drying.
Scale-up and process development. Modelling of processes with mathematical and statistical models.
Optimisation and Process Development
Design of Experiments (DoE), optimisation of plants during ongoing production, process simulations
Process Analytical Technology (PAT)
Linking experimental data to models, using these models for process development and optimisation
Finding robust production parameters. This is where manufacturing profitability lies.
Quality Management under GMP (Pharmaceuticals, Medical Technology, Chemicals, Industry) or ISO 9001, ISO 13485
We are dealing with the optimisation of warehouses, routes, buffers for future extensions and conversions.
Our project management also includes change management. This is the only way to achieve sustainable implementation.
Without implementation, no project helps.
Transfer of technologies, productions, expansion and refurbishment of facilities
Transfer of analytical methods under GMP, including validations, verifications with all necessary documentation
Professional process optimisations (in collaboration with Japanese specialists).
These are the processes and tools we use the most
Scale-up and process development.
Bespoke software development for tailored solutions
Development of freeze-drying cycles. Scale up & Scale down. Technology transfer of freeze-drying.
Scale-up and process development. Modelling of processes with mathematical and statistical models.
Optimisation and Process Development
Design of Experiments (DoE), optimisation of plants during ongoing production, process simulations
Process Analytical Technology (PAT)
Linking experimental data to models, using these models for process development and optimisation
Finding robust production parameters. This is where manufacturing profitability lies.
Quality Management under GMP (Pharmaceuticals, Medical Technology, Chemicals, Industry) or ISO 9001, ISO 13485
We are dealing with the optimisation of warehouses, routes, buffers for future extensions and conversions.
Our project management also includes change management. This is the only way to achieve sustainable implementation.
Without implementation, no project helps.
Transfer of technologies, productions, expansion and refurbishment of facilities
Transfer of analytical methods under GMP, including validations, verifications with all necessary documentation
Professional process optimisations (in collaboration with Japanese specialists).
The performance of CMOs and CDMOs improves through clear governance, transparent key performance indicators (KPIs), and active management of external partners. Binding targets for supply capability, quality, costs, deviations, and response times are important. Without structured control, delays, quality problems, and rising costs often occur.
Pharma supply chains become weak when external partners are not sufficiently managed, risks are identified too late, and quality or delivery issues are only addressed reactively. Often, robust key performance indicators, clear responsibilities, and an active escalation structure are lacking. This leads to stock shortages, delivery delays, cost increases, and compliance risks.
Costs can be reduced with CMOs by making the real cost drivers transparent and actively managing them. These include unnecessary inventory, special promotions, rework, inefficient processes, quality issues, and unstable delivery schedules. The goal is not pure price pressure, but a more stable and economical collaboration with the CMO.
External support makes sense when CMOs repeatedly deliver late, costs rise, quality issues occur, or internal teams lack sufficient transparency regarding actual performance. Experienced experts help identify risks, structure escalations, and regain control over external partners.
Third-party management improves through clear responsibilities, regular performance measurement, structured communication, and consistent risk management. The connection between quality, supply chain, procurement, QA, and CMO control is particularly important. This way, external partners are not just managed but actively led.
Warning signs include repeated delivery delays, rising costs, unclear root cause analyses, frequent quality issues, and a lack of transparency on the actual status. If decisions are largely reactive and internal teams are chasing the CMO, control is already weakened.